Do you want to know more about AMR?
Infobites
Antimicrobial resistance now a leading cause of death worldwide, study finds
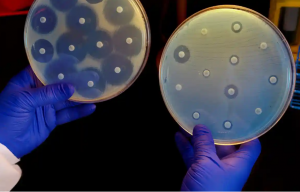 Antimicrobial resistance poses a significant threat to humanity, health leaders have warned, as a study reveals it has become a leading cause of death worldwide and is killing about 3,500 people every day.
Antimicrobial resistance poses a significant threat to humanity, health leaders have warned, as a study reveals it has become a leading cause of death worldwide and is killing about 3,500 people every day.
More than 1.2 million – and potentially millions more – died in 2019 as a direct result of antibiotic-resistant bacterial infections, according to the most comprehensive estimate to date of the global impact of antimicrobial resistance (AMR).
The stark analysis covering more than 200 countries and territories was published in the Lancet. It says AMR is killing more people than HIV/Aids or malaria. Many hundreds of thousands of deaths are occurring due to common, previously treatable infections, the study says, because bacteria that cause them have become resistant to treatment.
“These new data reveal the true scale of antimicrobial resistance worldwide, and are a clear signal that we must act now to combat the threat,” said the report’s co-author Prof Chris Murray, of the Institute for Health Metrics and Evaluation at the University of Washington.
https://www.theguardian.com/society/2022/jan/20/antimicrobial-resistance-antibiotic-resistant-bacterial-infections-deaths-lancet-study
Is bacterial acidity a key to tackle antimicrobial resistance?
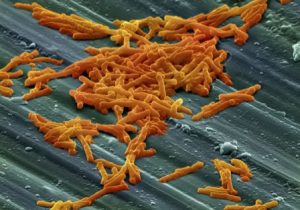 Decreasing bacterial acidity could help reduce antimicrobial resistance by eliminating bacteria that can survive being treated with antibiotics. Scientists at have developed a novel method, which allows users to measure the pH of individual bacteria before, during and after treatment with antibiotics.
Decreasing bacterial acidity could help reduce antimicrobial resistance by eliminating bacteria that can survive being treated with antibiotics. Scientists at have developed a novel method, which allows users to measure the pH of individual bacteria before, during and after treatment with antibiotics.
https://www.pharmamicroresources.com/2021/09/is-bacterial-acidity-key-to-tackle.html?m=1
Antibiotic Footprint – Interactive map
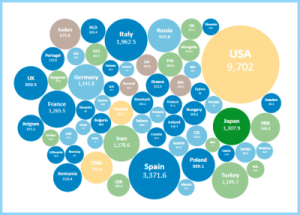 This website presents the total amount of antibiotics consume around the world
This website presents the total amount of antibiotics consume around the world
What is Antibiotic Footprint?
Antibiotic Footprint is a tool to measure and understand the total magnitude of antibiotic use. Using a holistic approach, Antibiotic Footprint seeks to measure the total amount of antibiotics used in all human activities, including direct antibiotics consumption at the community and hospital levels, and indirect consumption in agricultural production.
https://www.antibioticfootprint.net/
Deadly Contact: Nanoparticles Combat Antibiotic-Resistant Bacteria
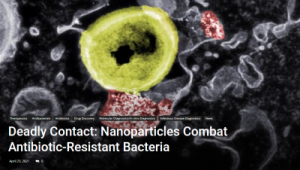 Nanoparticles are a relatively new competitor in the fight against antibiotic-resistant bacteria. A team of researchers from Swiss Federal Laboratories for Materials Science and Technology (Empa) and ETH Zurich is developing nanoparticles that use a completely different mode of action from conventional antibiotics. While antibiotics have difficulty in penetrating human cells, nanoparticles, due to their small size and structure, can penetrate the membrane of affected cells. Because of that, they play a role in combatting intracellular pathogens, like multi-resistant staphylococci (MRSA).
Nanoparticles are a relatively new competitor in the fight against antibiotic-resistant bacteria. A team of researchers from Swiss Federal Laboratories for Materials Science and Technology (Empa) and ETH Zurich is developing nanoparticles that use a completely different mode of action from conventional antibiotics. While antibiotics have difficulty in penetrating human cells, nanoparticles, due to their small size and structure, can penetrate the membrane of affected cells. Because of that, they play a role in combatting intracellular pathogens, like multi-resistant staphylococci (MRSA).
https://www.genengnews.com/news/deadly-contact-nanoparticles-combat-antibiotic-resistant-bacteria/
New study shows microplastics turn into ‘hubs’ for pathogens, antibiotic-resistant bacteria
 The ultra-fine plastic particles, less than 5 millimetres in length, found in everything from cosmetics, toothpaste and clothing microfibers, to our food, air and drinking water.
The ultra-fine plastic particles, less than 5 millimetres in length, found in everything from cosmetics, toothpaste and clothing microfibers, to our food, air and drinking water.
Now, researchers at New Jersey Institute of Technology have shown that ubiquitous microplastics can become ‘hubs’ for antibiotic-resistant bacteria and pathogens to grow once they wash down household drains and enter wastewater treatment plants—forming a slimy layer of buildup, or biofilm, on their surface that allows pathogenic microorganisms and antibiotic waste to attach and comingle.
https://phys-org.cdn.ampproject.org/c/s/phys.org/news/2021-03-microplastics-hubs-pathogens-antibiotic-resistant-bacteria.amp
Could soil used in ancient Irish folk medicine fight antimicrobial resistance?
 In a new discovery, a team of scientists have shown that Northern Irish soil has antibiotic-producing bacteria that could help in the fight against antimicrobial resistance.
In a new discovery, a team of scientists have shown that Northern Irish soil has antibiotic-producing bacteria that could help in the fight against antimicrobial resistance.
The Traditional Medicine Group, which is a collaboration between scientists at Swansea University, and researchers in Brazil and Ireland, has discovered an antibiotic-producing species in Irish soil, and believe they have identified new varieties of antibiotics that could help to save lives.
https://www.healtheuropa.eu/could-soil-used-in-ancient-irish-folk-medicine-fight-antimicrobial-resistance/104970/
May consumption of probiotic food have an adverse effect, besides the health benefits?
 A glimpse of antimicrobial resistance gene diversity in kefir and yoghurt
A glimpse of antimicrobial resistance gene diversity in kefir and yoghurt
Fermented foods, like yoghurt and kefir, naturally contains bacteria in high amounts.
New research has found numerous antimicrobial resistance genes (ARGs) in commonly used fermenting bacteria.
https://www.nature.com/articles/s41598-020-80444-5
Livestock Farming’s Role In The Antibiotic Resistance Crisis
 Public Health England recently announced that there are 165 new antibiotic-resistant infections every day in England. Antimicrobial-resistance (AMR) is a global problem and it is increasing. Where are these new infections coming from and should we be worried?
Public Health England recently announced that there are 165 new antibiotic-resistant infections every day in England. Antimicrobial-resistance (AMR) is a global problem and it is increasing. Where are these new infections coming from and should we be worried?
https://plantbasednews.org/opinion/livestock-farming-antibiotic-resistance-crisis/
COVID and AMR: Crisis Reframes Antibiotics Fight
 2020 will be long-remembered for the COVID-19 pandemic, which has taken almost 1.5 million lives globally as of Dec. 1. However, another pandemic—drug-resistant bacterial and fungal infections—kills more than 700,000 people each year. Worse still, it is estimated that by 2050, well after SARS-CoV-2 has hopefully been defeated, 10 million people worldwide will die from antimicrobial-resistant (AMR) infections.
2020 will be long-remembered for the COVID-19 pandemic, which has taken almost 1.5 million lives globally as of Dec. 1. However, another pandemic—drug-resistant bacterial and fungal infections—kills more than 700,000 people each year. Worse still, it is estimated that by 2050, well after SARS-CoV-2 has hopefully been defeated, 10 million people worldwide will die from antimicrobial-resistant (AMR) infections.
https://www.pharmexec.com/view/covid-and-amr-crisis-reframes-antibiotics-fight
4 Key Priorities for Fighting Superbugs in 2021
 Pandemic offers lessons for combating antibiotic resistance, underscores need for greater public health preparedness
Pandemic offers lessons for combating antibiotic resistance, underscores need for greater public health preparedness
Over the past year, COVID-19 has taken a grave toll in lives as well as on medical and health care systems worldwide. The pandemic has laid bare the importance of public health readiness and the myriad consequences when such a crisis strikes an unprepared population.
Even as the world is working to defeat COVID-19, another global health threat is lurking: antibiotic resistance.
https://www.pewtrusts.org/en/research-and-analysis/articles/2021/01/14/4-key-priorities-for-fighting-superbugs-in-2021
The surfaces that kill bacteria and viruses
 By copying the texture of insect wings or using new types of materials to create surfaces that kill or inhibit microbes, we could stop infections before they even get into the body.
By copying the texture of insect wings or using new types of materials to create surfaces that kill or inhibit microbes, we could stop infections before they even get into the body.
Currently, 700,000 people die each year of drug-resistant diseases.
As Larrouy-Maumus, an infectious disease researcher at Imperial College London in the UK, warns, “If we do nothing, 10 million people per year will die.”
He is among those looking for new ways to tackle antimicrobial resistance. His plan is to turn the very surfaces that many of these pathogens use to spread from person to person into weapons against them.
https://www.bbc.com/future/article/20200529-the-surfaces-that-kill-bacteria-and-viruses
A New Type of Antibiotics Help the Immune System Fight Pathogens
 Reporting in Nature, scientists have identified a new group of compounds that may help us get out of the antibiotic-resistant bacteria crisis. These novel drugs work by improving the immune system’s natural ability to fight pathogens while also battling the infection directly.
Reporting in Nature, scientists have identified a new group of compounds that may help us get out of the antibiotic-resistant bacteria crisis. These novel drugs work by improving the immune system’s natural ability to fight pathogens while also battling the infection directly.
https://www.labroots.com/trending/microbiology/19530/type-antibiotics-help-immune-system-fight-pathogens
AMR, the next pandemic?
 It is now estimated 700,000 people die each year as a result of drug-resistant diseases, with this number predicted to increase to over 10 million deaths per year by 2050. The current COVID-19 pandemic appears to have accelerated the threat of antimicrobial resistance (AMR), as many patients admitted to hospitals displaying COVID-19 symptoms are treated with antibiotics to reduce their chances of contracting secondary bacterial infections, making resistant bacteria more common.
It is now estimated 700,000 people die each year as a result of drug-resistant diseases, with this number predicted to increase to over 10 million deaths per year by 2050. The current COVID-19 pandemic appears to have accelerated the threat of antimicrobial resistance (AMR), as many patients admitted to hospitals displaying COVID-19 symptoms are treated with antibiotics to reduce their chances of contracting secondary bacterial infections, making resistant bacteria more common.
https://microbiologysociety.org/blog/antimicrobial-resistance-the-next-pandemic.html
Dealing with antimicrobial resistance during COVID-19
 To coincide with Antimicrobial Awareness Week 2020, a new report of case studies has been published that gives a platform to people living with drug-resistant infections, those that have survived them, and to the clinicians contending with them.
To coincide with Antimicrobial Awareness Week 2020, a new report of case studies has been published that gives a platform to people living with drug-resistant infections, those that have survived them, and to the clinicians contending with them.
The report, from the frontlines of the pandemic in the UK, US, South Africa and India, captures unique views on the COVID-19 pandemic, providing a first-hand look at how the real-life experience and challenges of antimicrobial resistance (AMR) collide with the realities of COVID-19.
The report is a warning that without action on the slow-moving pandemic of AMR, more lives will be lost and the fundamentals of modern medicine will break down.
https://www.healtheuropa.eu/dealing-with-antimicrobial-resistance-during-covid-19/104085/
AMR: what’s the issue, and why is it so important?
 AMR causes 700,000 deaths every year and the numbers keep growing. Antimicrobials are drugs used against different kinds of viruses, bacteria, etc.; resistance is developed through exposure to these drugs or the misuse of them. But why are antimicrobials important?
AMR causes 700,000 deaths every year and the numbers keep growing. Antimicrobials are drugs used against different kinds of viruses, bacteria, etc.; resistance is developed through exposure to these drugs or the misuse of them. But why are antimicrobials important?
Learn more about the disease and the global situation here:
https://www.thebureauinvestigates.com/stories/2016-02-07/antimicrobial-resistance-whats-the-issue-and-why-is-it-so-important
How do we reduce antibiotic resistance from livestock?
 AMR can be a source of danger for both animals and human beings. Animals are also prone to infections and antibiotics are a treatment solution for the infected ones. However, the mass use of antibiotics to protect the livestock’s health causes serious issues.
AMR can be a source of danger for both animals and human beings. Animals are also prone to infections and antibiotics are a treatment solution for the infected ones. However, the mass use of antibiotics to protect the livestock’s health causes serious issues.
How does AMR work when it comes to animals? When and how to use antibiotics for livestock? Click here to find out more:
https://ourworldindata.org/antibiotic-resistance-from-livestock
Frontiers 2017: Emerging Issues of Environmental Concern
 AMR is one of the biggest health issues of the 21st century – but what else is it threatening? The unnecessary use of antibiotics has created other problems such as the careless disposal of antimicrobials. Now, this is yet another environmental issue facing our planet.
AMR is one of the biggest health issues of the 21st century – but what else is it threatening? The unnecessary use of antibiotics has created other problems such as the careless disposal of antimicrobials. Now, this is yet another environmental issue facing our planet.
How does the careless disposal of antimicrobials produce bacteria that can resist them? How does it affect the environment? How can we fight against it? Read more about the topic:
https://www.unenvironment.org/resources/frontiers-2017-emerging-issues-environmental-concern
AMR: a major European and global challenge
 If current trends continue, we will revert to world where simple infections are no longer treatable. See European Commission’s fact sheet about AMR:
If current trends continue, we will revert to world where simple infections are no longer treatable. See European Commission’s fact sheet about AMR:
https://ec.europa.eu/health/amr/sites/health/files/antimicrobial_resistance/docs/amr_2017_factsheet.pdf
Antimicrobial resistance (AMR): definition by WHO
 What is Antimicrobial resistance? Antimicrobial resistance happens when microorganisms (such as bacteria, fungi, viruses, and parasites) change when they are exposed to antimicrobial drugs (such as antibiotics, antifungals, antivirals, antimalarials, and anthelmintics). Microorganisms that develop antimicrobial resistance are sometimes referred to as “superbugs” .
What is Antimicrobial resistance? Antimicrobial resistance happens when microorganisms (such as bacteria, fungi, viruses, and parasites) change when they are exposed to antimicrobial drugs (such as antibiotics, antifungals, antivirals, antimalarials, and anthelmintics). Microorganisms that develop antimicrobial resistance are sometimes referred to as “superbugs” .
https://www.who.int/health-topics/antimicrobial-resistance
The economic burden of AMR
 The world needs accurate estimates of the burden of AMR threat in terms of both health and cost, and to parameterise cost-effectiveness evaluations of interventions aimed at solving this problem. See the WHO fact sheet for more information:
The world needs accurate estimates of the burden of AMR threat in terms of both health and cost, and to parameterise cost-effectiveness evaluations of interventions aimed at solving this problem. See the WHO fact sheet for more information:
https://www.who.int/infection-prevention/tools/IPC_AMR_A4.pdf
What would a world without antibiotics be like?
 It’s sometimes called “the silent pandemic”. The video explores the growing problem of antibiotic resistance – and what can be done about it.
It’s sometimes called “the silent pandemic”. The video explores the growing problem of antibiotic resistance – and what can be done about it.
https://www.bbc.co.uk/ideas/videos/what-would-a-world-without-antibiotics-be-like/p0cgmypx
Are phages more effective than antibiotics?
In recent years bacteriophages have gained popularity on being the last option when antibiotics have failed. Many people are still doubting the safety of using bacteriophages for clinical application due to fear of some imaginable implications basing on the fact that these are viral particles introduced to the human body. In some scenarios, cases of multidrug-resistant strains of bacteria were successfully treated using bacteriophage while in most of the cases no side effect was directly related to the phage therapy offered to the patient.
https://www.thephage.xyz/2021/07/are-phages-more-effective-than.html
Phage therapy as a potential solution in the fight against AMR: obstacles and possible futures
 Phage therapy, the use of bacteriophage viruses to treat bacterial infections, has existed for more than a hundred years. However, the practice is struggling to develop, despite growing support over the past 15 years from researchers and doctors, who see it as a promising therapy in the context of the rise of antimicrobial resistance (AMR). This paper aims to show that antibiotics also form a kind of epistemological infrastructure, which acts as a powerful inhibitor to the development of phage therapy. In this sense antibiotics prevent the development of solutions to the problem they contribute to create.
Phage therapy, the use of bacteriophage viruses to treat bacterial infections, has existed for more than a hundred years. However, the practice is struggling to develop, despite growing support over the past 15 years from researchers and doctors, who see it as a promising therapy in the context of the rise of antimicrobial resistance (AMR). This paper aims to show that antibiotics also form a kind of epistemological infrastructure, which acts as a powerful inhibitor to the development of phage therapy. In this sense antibiotics prevent the development of solutions to the problem they contribute to create.
https://www.nature.com/articles/s41599-020-0478-4
New research: Dog food sold across Europe contains antibiotic-resistant bacteria
 A new research presented at the European Congress of Clinical Microbiology & Infectious Diseases (ECCMID) reveals that raw dog food is a significant source of antibiotic-resistant bacteria, making it an international public health risk.
A new research presented at the European Congress of Clinical Microbiology & Infectious Diseases (ECCMID) reveals that raw dog food is a significant source of antibiotic-resistant bacteria, making it an international public health risk.
Some of the multidrug-resistant bacteria in raw dog food were found to be identical to those found in hospital patients in several different European countries. Raw dog food presents an international public health risk due to antibiotic-resistant bacteria in a range of different types of raw dog food.
https://ask-bioexpert.com/blog-post/new-research-dog-food-sold-across-europe-contains-antibiotic-resistant-bacteria/
Deforestation in the Amazon increases diversity of antibiotic-resistant bacteria
 In Brazil, a study conducted by researchers affiliated with the University of São Paulo (USP) and collaborators showed that deforestation in the Amazon causes an increase in the diversity of antibiotic-resistant bacteria.
In Brazil, a study conducted by researchers affiliated with the University of São Paulo (USP) and collaborators showed that deforestation in the Amazon causes an increase in the diversity of antibiotic-resistant bacteria.
Bacteria produce substances with which to attack each other in a competition for resources that’s usual in any environment. When an area is deforested, however, several factors intensify this competition, favoring the bacteria that can resist these substances. If they reach humans, these microorganisms can become a major problem.
https://www.news-medical.net/news/20210312/Deforestation-in-the-Amazon-increases-diversity-of-antibiotic-resistant-bacteria.aspx
New model to predict how bacteria develop antibiotic resistance
 Using theoretical models of bacterial metabolism and reproduction, scientists can predict the type of resistance that bacteria will develop when they are exposed to antibiotics. This has now been shown by an Uppsala University research team, in collaboration with colleagues in Cologne, Germany. The study is published in the journal Nature Ecology and Evolution.
Using theoretical models of bacterial metabolism and reproduction, scientists can predict the type of resistance that bacteria will develop when they are exposed to antibiotics. This has now been shown by an Uppsala University research team, in collaboration with colleagues in Cologne, Germany. The study is published in the journal Nature Ecology and Evolution.
https://www.pharmamicroresources.com/2021/03/new-model-to-predict-how-bacteria.html?m=1
Now, more than ever, we need to commit to fighting the spread of AMR
 Imagine that 2020, with all its upheaval, becomes the norm in a future where we will have lost the fight against antimicrobial resistance.
Imagine that 2020, with all its upheaval, becomes the norm in a future where we will have lost the fight against antimicrobial resistance.
Fighting AMR is not an easy task, and the current COVID-19 pandemic has only made things worse.
The lessons learned during the Coronavirus crisis will be invaluable in better coping with the prevention and control of future health crises, such as AMR.
https://www.theparliamentmagazine.eu/news/article/the-silent-pandemic
Antibiotic resistance in ophthalmology is an urgent global health priority
 Cataract surgery, where prophylactic antibiotics are used, is Europe’s most frequently performed surgery. Ophthalmologists have a central role to play managing this urgent global health concern.
Cataract surgery, where prophylactic antibiotics are used, is Europe’s most frequently performed surgery. Ophthalmologists have a central role to play managing this urgent global health concern.
https://jointherevolution.santen.eu/?gclid=CjwKCAiAl4WABhAJEiwATUnEF7zahl9YwJMjZfE4pGiqbR-FmSLhT0Y8STLTxDVHkowF_YzvXhR_DhoCD6sQAvD_BwE
Lurching from one pandemic to another? From the frontlines of antibiotic resistance during Covid-19
 A new report of case studies published to coincide with World Antimicrobial Awareness Week 2020 gives a platform to people living with drug-resistant infections, those that have survived them, and to the clinicians contending with them, capturing their unique views on the Covid-19 pandemic.
A new report of case studies published to coincide with World Antimicrobial Awareness Week 2020 gives a platform to people living with drug-resistant infections, those that have survived them, and to the clinicians contending with them, capturing their unique views on the Covid-19 pandemic.
https://indusdictum.com/2020/12/18/lurching-from-one-pandemic-to-another-from-the-frontlines-of-antibiotic-resistance-during-covid-19/
Can we change our behaviour to prevent superbugs?
 In some farming sectors, antibiotics are used routinely to maintain animal health during the year.
In some farming sectors, antibiotics are used routinely to maintain animal health during the year.
The invisible threat of antibiotic resistance is a health issue that urgently needs addressing by all of us, from farmers to consumers.
The WHO reported that reducing antibiotic use in farm animals could reduce antibiotic-resistant bacteria in these animals by up to 39% – good news for them and us.
https://www.rte.ie/brainstorm/2021/0107/1188209-antibiotic-resistance-superbugs-farming-eu/
Scientists pave way to reducing antibiotic resistance build-up in waterways
 Around 70 per cent of the antibiotics we take as medicine ends up in the natural environment, through trace residues excreted by patients and inappropriate disposal of medicines, among other sources. Bacteria are also present in these wastewaters, and when they are exposed to antibiotics they can evolve resistance within these environments. This could mean an increased threat to human health, if resistant bacteria enter and colonise the gut, for example through swallowing water while swimming. The bacteria could then transfer resistance to human-associated bacteria, meaning antibiotics are less likely to work when they are needed…
Around 70 per cent of the antibiotics we take as medicine ends up in the natural environment, through trace residues excreted by patients and inappropriate disposal of medicines, among other sources. Bacteria are also present in these wastewaters, and when they are exposed to antibiotics they can evolve resistance within these environments. This could mean an increased threat to human health, if resistant bacteria enter and colonise the gut, for example through swallowing water while swimming. The bacteria could then transfer resistance to human-associated bacteria, meaning antibiotics are less likely to work when they are needed…
https://phys-org.cdn.ampproject.org/c/s/phys.org/news/2020-10-scientists-pave-antibiotic-resistance-build-up.amp
Antimicrobial resistance could get worse during the coronavirus pandemic
 More COVID-19 patients are getting antibiotics when they don’t need them, and this could make the situation worse.
More COVID-19 patients are getting antibiotics when they don’t need them, and this could make the situation worse.
https://thehill-com.cdn.ampproject.org/c/s/thehill.com/changing-america/well-being/prevention-cures/521455-antimicrobial-resistance-could-get-worse-during?amp
What Is Antibiotic Resistance?
 Did you know that over 70% of antibiotics produced globally are not given to sick humans, but farm animals? This allows them to grow at unnatural rates, and live through the conditions of industrial animal agriculture.
Did you know that over 70% of antibiotics produced globally are not given to sick humans, but farm animals? This allows them to grow at unnatural rates, and live through the conditions of industrial animal agriculture.
“The world is heading towards a post-antibiotic era in which common infection”
This clip was taken from our film ‘Vegan 2020’ available to watch now on our YouTube…
60 seconds video on Instagram:
https://www.instagram.com/tv/CJrDRO_hfid/?igshid=ofyy1vejqfqa
Powerful antibiotics discovered using AI
 Machine learning spots molecules that work even against ‘untreatable’ strains of bacteria.
Machine learning spots molecules that work even against ‘untreatable’ strains of bacteria.
A pioneering machine-learning approach has identified powerful new types of antibiotic from a pool of more than 100 million molecules — including one that works against a wide range of bacteria, including tuberculosis and strains considered untreatable.
https://www.nature.com/articles/d41586-020-00018-3
We need new antimicrobials to prevent the next infectious disease crisis
 Imagine if scientists had seen Covid-19 coming years in advance yet did little to prepare. Unthinkable, right?
Imagine if scientists had seen Covid-19 coming years in advance yet did little to prepare. Unthinkable, right?
Yet that’s exactly what’s happening with another infectious disease crisis — the one caused by antibiotic-resistant bacteria and fungi. So-called superbugs already kill more than 700,000 people each year. And the World Health Organization warns that by 2050 the annual death toll could reach 10 million if we don’t use the time to get prepared.
The antibiotics and other antimicrobial drugs needed to prevent such a calamity don’t yet exist — and they’re years away from patients.
https://www.statnews.com/2020/08/12/we-need-new-antimicrobials-to-prevent-the-next-infectious-disease-crisis/
Global Commitment to Diagnostic Tests to Fight Antimicrobial Resistance
 AMR has become a top priority due to the possible global health and economic crisis that it has the potential to cause. Identifying the organisms causing the infection can help the AMR situation; diagnostics and diagnostic tests are the keys to achieving this.
AMR has become a top priority due to the possible global health and economic crisis that it has the potential to cause. Identifying the organisms causing the infection can help the AMR situation; diagnostics and diagnostic tests are the keys to achieving this.
How do the tests work? How can they be transferred into the system?
http://amr.bd.com/assets/pdf/8%20Global%20Commitment%20on%20Diagnostic%20Tests.pdf
Antimicrobial resistance
 AMR occurs when the medicine used to treat a person’s condition becomes ineffective and, worldwide, this has influenced the treatment options for common infectious diseases. It is fast becoming one of the greatest threats to global health.
AMR occurs when the medicine used to treat a person’s condition becomes ineffective and, worldwide, this has influenced the treatment options for common infectious diseases. It is fast becoming one of the greatest threats to global health.
How does the resistance affect the body or other diseases and how does WHO see the issue? See more information here:
https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance
AMR and diagnostics: pointing the way to better infection control
 AMR has a long history: evidence of antibiotic resistance genes dates back to 10,000 and 2 billion years ago as a response to natural antibiotics. Giving pharmacists a bigger role and improving diagnostics are some of the ways we can reduce the unnecessary use of antibiotics.
AMR has a long history: evidence of antibiotic resistance genes dates back to 10,000 and 2 billion years ago as a response to natural antibiotics. Giving pharmacists a bigger role and improving diagnostics are some of the ways we can reduce the unnecessary use of antibiotics.
What changed over the years that made it one of the biggest threats of the 21st century? How can diagnostics processes and pharmacists help solve the problem?
https://www.pharmaceutical-journal.com/news-and-analysis/amr-and-diagnostics-pointing-the-way-to-better-infection-control/20203597.article
Effective IPC saves money!
 Effective infection prevention and control (IPC) makes sense from a fiscal point of view, according to this UK National Institute for Health and Care Excellence (NICE) briefing paper:
Effective infection prevention and control (IPC) makes sense from a fiscal point of view, according to this UK National Institute for Health and Care Excellence (NICE) briefing paper:
https://www.nice.org.uk/guidance/qs61/documents/infection-control-briefing-paper2

